- Mitsubishi Chemical Corporation
- Ion Exchange Resin Business Gr. Separation Materials Dept.
Strongly Acidic Cation Exchange ResinsDIAION™ sereies
Mitsubishi Chemical Corporation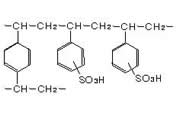
Characteristics
[Close]
The strongly acidic cation exchange resins are bead-like products which have a sulfonic acid group in the cross-linked styrene frame.
The sulfonic acid group (-HSO3) is the exchange group and behaves like a strong acid, dissociating to (-SO3)- and H+ even in acidic solutions, not to mention in alkaline solutions.
There are a gel type (SK series), a porous type (PK series), and a uniform bead size type (UBK series).


An ion exchange reaction is simply a pair of ions changing places. The reactions shown below illustrate this process for a strongly acidic cation exchange resin. Salts such as NaCl and CaCl2 or a base such as NaOH can undergo the ion exchange reaction when exposed to the resin in solution.
R-SO3H + NaCl → R-SO3Na + HCl
2R-SO3H + CaCl2→ (R-SO3)2Ca + 2HCl
R-SO3H + NaOH → R-SO3Na + H2O
(R represents the ion exchange resin matrix.)
Although the strongly acidic cation exchange resin adsorbs all positive ions, the adsorption strength depends on the type of ion. The index of this strength is called selectivity. In general, selectivity will increase as the ion valence increases. (For example, Na+ < Ca++ < Al+++ < Th++++.) For the same valence, selectivity increases as atomic number increases. (Li+ < Na+ < K+ <Rb+ < Cs+, Mg++ <Ca++ < Sr++ <Ba++)
These DIAION™ Series resins can be used across the full pH range (0-14), and are relatively stable to temperature, even withstanding high temperatures of 100-120℃.
Applications
[Close]
They are used in a wide variety of fields including water purification, water softening, wastewater treatment, purification of pharmaceuticals and food, as well as catalysis.
Usage
Packed column or Batch processing
Lineup / Specifications
[Close]
Refer to the "ION EXCHANGE RESINS "[別窓表示] websites.
Inquiries Concerning Products
View the products of Separation Materials Dept., Mitsubishi Chemical[Open in a new window]



