Multilayer Co-extruded Film for Medical Supplies and Pharmaceuticals PackagingDIAMIRON™
Mitsubishi Chemical Corporation- Language
We are able to create co-extruded films by using our unique co-extrusion technology to merge multiple resins together in a die.
This high-performance film inherits the properties of each resin used.
We can customize the properties of the film with functionality based on your needs, such as producing a film with high barrier properties.
We constructed a special, clean production environment to produce DIAMIRON as an extremely high-quality primary container. This material is used in such products as transfusion and infusion bags and sterile blister packs for medical supplies.
DIAMIRON is produced at the Azai Plant under ISO 9001 and FSSC 22000-compliant quality control.
Visit the product website for more information.
Applications
- Sterile blister packs for medical supplies
- Transfusion bags
Film for Sterile Blister Packs for Medical SuppliesDIAMIRON™ MF

DIAMIRON MF is a non-oriented, extruded multilayer film used mainly for blister packs. This product is sold in rolls of flat film. Multiple resins having different properties are extruded to produce this product. Our lineup includes many varieties from flexible film sheets to rigid sheets to meet any type of requirement. We also accept custom orders with short lead times if you would like to change the material configuration, thickness, or functionality to meet specific requirements such as compatibility with a particular sterilization method, packaging machine, or other fabrication conditions.
Characteristics
- Suitable moldability, transparency, and gloss properties.
- Excellent sealing properties with non-coated Tyvek® and sterilization paper. Opens easily once sealed.
- Compatible with EOG, radiation, autoclave, and other sterilization methods.
- Can be produced with properties in accordance with specific requirements. Some examples include easy-opening property, resistance to pinholes, aroma retention, chemical resistance, moistureproof properties, oxygen barrier properties, and so on.
- *Tyvek is a registered trademark of DuPont.
Applications
- Syringes
- Surgical kit products
- Prefilled syringes
- Catheters
- Drapes
- Gauze
- Cotton swabs
- Cotton balls
Lineup
| Type | Basic configuration | Recommended cover material | Features | Sterilization method | ||
|---|---|---|---|---|---|---|
| EOG | Radiation | Autoclave |
||||
| C1 | NY/PE | PET/PE, Coated Tyvek, Coated sterilization paper |
Gloss, moldability | O | O | X |
| C4 | NY/PE(EP) | PET/PE, Non-coated Tyvek, Non-coated sterilization paper |
Gloss, moldability, easy-open |
O | O | X |
| NY/PP(EP) | PET/PP, Non-coated Tyvek, Non-coated sterilization paper |
Gloss, heat resistance, easy-open |
O | △ | O | |
| C5 | NY/PP | PET/PP, Coated Tyvek, Coated sterilization paper |
Gloss, heat resistance | O | △ | O |
| F1 | PE/NY/PE | PET/PE, Coated Tyvek, Coated sterilization paper |
Flexibility, moldability | O | O | X |
| F4 | PE/NY/PE(EP) | PET/PE, Non-coated Tyvek, Non-coated sterilization paper |
Flexibility, moldability, easy-open |
O | O | X |
| H6 | PP/NY/PE(EP) | PET/PE, Non-coated Tyvek, Non-coated sterilization paper |
Firm, moldability, easy-open |
O | △ | X |
| H4 | PP/NY/PP | PET/PP, Coated Tyvek, Coated sterilization paper |
Firm and heat resistance | O | △ | O |
| PP/NY/PP(EP) | PET/PP, Coated Tyvek, Coated sterilization paper |
Firm, heat resistance, easy-open |
O | △ | O | |
- * EP is an acronym for the easy-peel layer.
High strength Multilayer Film for Transfusion BagsDIAMIRON M™

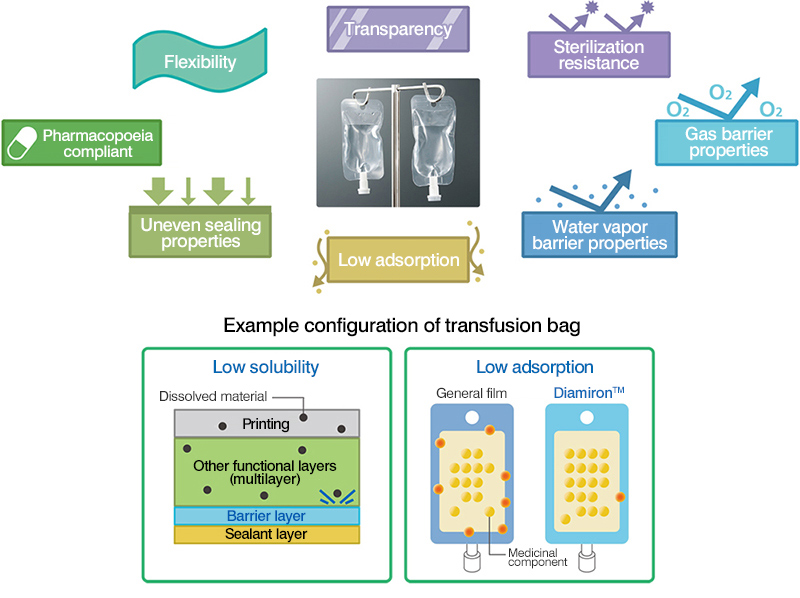
DIAMIRON M has a multilayer design that enables us to provide the performance and functionality required for transfusion and infusion bags. We constructed a special production environment to develop and produce Diamiron as an extremely high-quality primary container and film for transfusion bags.
Characteristics
- This film for liquid containers has excellent transparency, flexibility, and impact resistance.
- Can withstand high-temperature, high-pressure steam sterilization processes up to 121°C.
- Optimal for use in transfusion bags due to the extremely minimal adsorption and elution of active substances.
- This material complies with the Japanese Pharmacopoeia.
- This product is registered in the FDA Drug Master File.
- We produce this product under strict quality control including production in an ISO class 8 clean room.
- Can be produced with properties in accordance with specific requirements. Some examples include oxygen barrier properties, aroma retention, chemical resistance, moistureproof properties, peels easily, and so on.
If you would like to request any of the following materials, please write the name(s) of the document(s) on the inquiry form.
- Pharmaceutical and Medical Grade Diamiron
- Medical Product Lineup



