Polyvinyl alcohol (PVA,PVOH) for Pharmaceutical Excipients GOHSENOL™ EG
- Mitsubishi Chemical Corporation
- Food / Healthcare Dept.
These are links for moving within this page
Polyvinyl Alcohol is a pharmaceutical excipient and listed in major official compendia.
It is widely used in many countries including Japan, the United States, and Europe.
Our GOHSENOL™ EG conforms to pharmacopoeia excipients including JPE*
1,USP*2 and EP*3
[Close]
GOHSENOL™ EG series is Polyvinyl Alcohol obtained through purification process. Production facility, production management, and quality assurance all meet the criteria of self-imposed standards of GMP for pharmaceutical excipients.
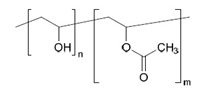
Degree of hydrolysis:{n/(m+n)}×100[mol%]
[Close]
| Grades | Degree of hydrolysis [mol%] |
Viscosity*1, *2(Unit of measurement) [mm2/s] |
|---|---|---|
| EG-03P | 86.5-89.0 | 3.0-3.8 (3.4) |
| EG-05P | 4.5 - 6.1 (5.3) | |
| EG-18P | 15.3- 20.7 (18.0) | |
| EG-22P | 19.0 - 25.6 (22.3) | |
| EG-30P | 25.5 - 34.5 (30.0) | |
| EG-40P | 36.6 - 49.4 (43.0) | |
| EG-48P | 41.3 - 55.7 (48.5) |
| Grades | Degree of hydrolysis [mol%] |
Viscosity*1, *2(Unit of measurement) [mm2/s] |
|---|---|---|
| EG-05PW | 86.5-89.0 | 4.5 - 6.1 (5.3) |
| EG-30PW | 25.5 - 34.5 (30.0) | |
| EG-40PW | 36.6 - 49.4 (43.0) |
[Close]
[Close]
[Close]

Polyvinyl alcohol (PVA,PVOH) for Pharmaceutical Excipients GOHSENOL™ EG
View the products of Food / Healthcare Dept., Mitsubishi Chemical[Open in a new window]
This is the end of this page